
by mandy | Jun 22, 2020 | Diet, Evidence Based, Nutrition, Science-Based
I was interested in learning more about vitamin E supplementation to address oxidative stress. Dr. Tan wrote a nice summary of Vitamin E in his book “The Truth about Vitamin E”. Vitamin E is a family of eight separate but related molecules, and that includes four tocopherols (delta, gamma, alpha and beta) and four tocotrienols [...]
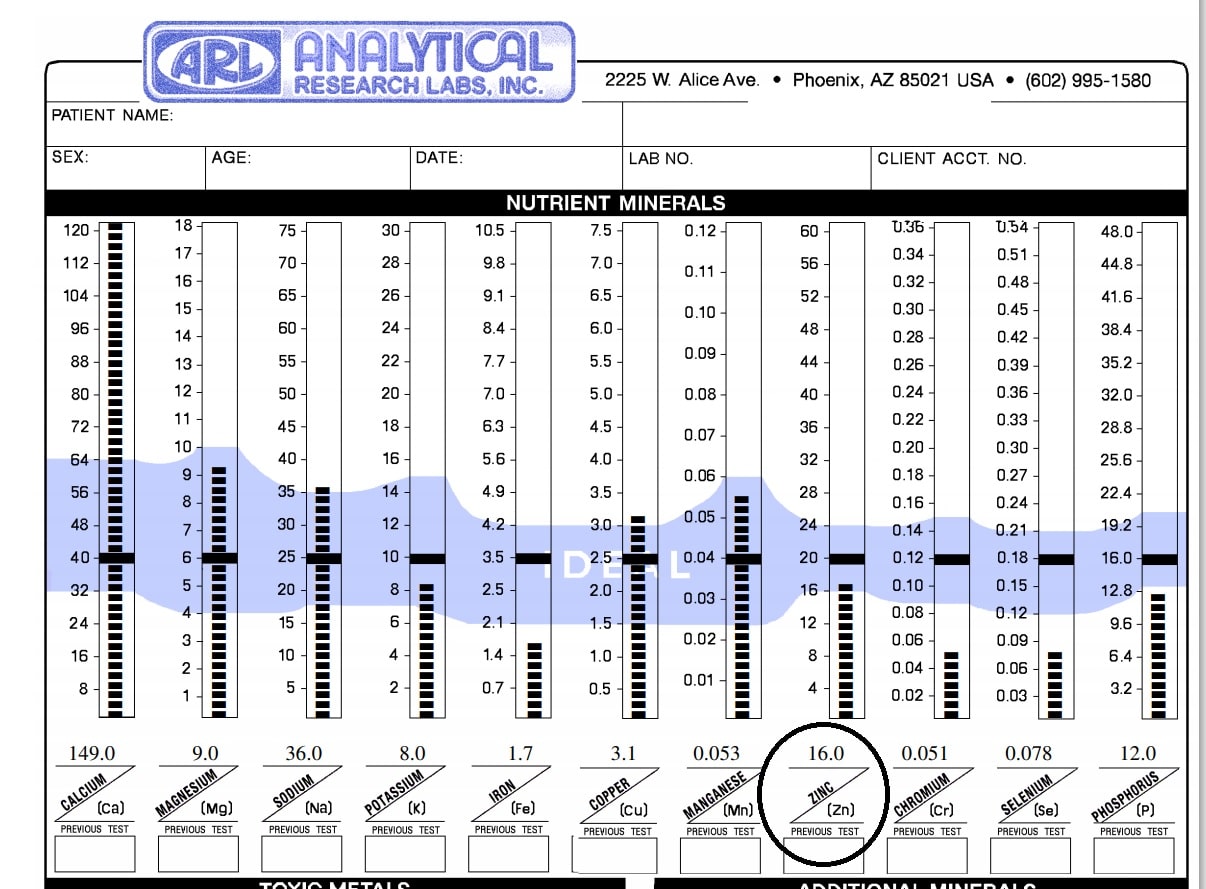
by mandy | Jun 22, 2020 | Diet, Evidence Based, Nutrition, Science-Based
Zinc is a very interesting mineral. It is also one I commonly see low in hair tissue mineral analysis (HTMA). It plays an important role in facilitating hundreds of biochemical reactions. Due to its role in enzymatic function, it can impact metabolic pathways such as carbohydrate, protein, nucleic acid, and lipid metabolism. It is also [...]

by mandy | Jun 22, 2020 | Evidence Based, Inflammation, Science-Based, Wellness
The relationship between thiamine and diabetes mellitus (DM) has been reported in the literature (Luong & Nguyen, 2012). Thiamine acts as a coenzyme for transketolase (Tk) and for the pyruvate dehydrogenase (PDH) and α-ketoglutarate dehydrogenase complexes. These enzymes play a fundamental role for intracellular glucose metabolism by increasing Krebs cycle activity (Luong & Nguyen, 2012). [...]

by mandy | Jun 15, 2020 | Evidence Based, Exercise, Science-Based, Wellness
It is no secret that exercise is very important for optimal health. But many people still do not see the connection and it seems to be a low priority for them when it comes to managing their health. However, I realize that many people truly do not understand how important it really is, even from [...]

by mandy | Jun 7, 2020 | Evidence Based, Exercise, Inflammation, Mental Health, Science-Based, Wellness
Having worked in the fitness industry for over 15 years and helping people with their weight loss goals, I always wondered what role exercise had on inflammation and aging. In obesity, various mechanisms are thought to contribute to a low- grade inflammation within the fat tissue affecting the development of several secondary diseases of aging [...]






