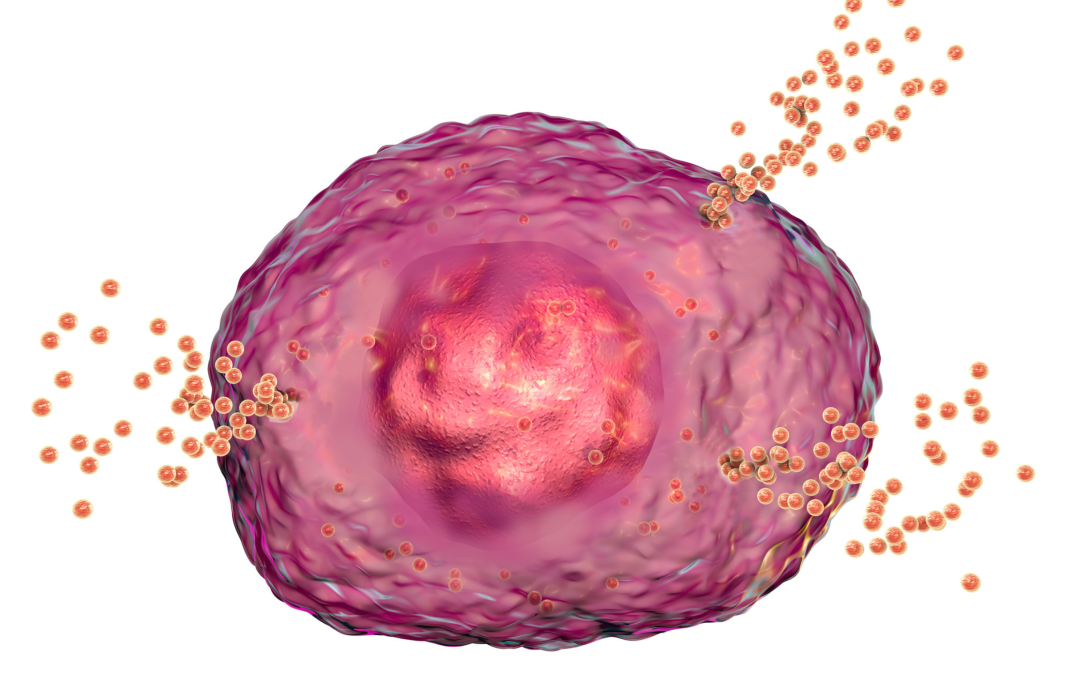
by mandy | Mar 24, 2024 | Detox, Evidence Based, Genetics, Inflammation, Nutrients, Nutrition, Science-Based, Supplements
The intricate interactions within our body's immune system, particularly involving mast cells, play a crucial role in inflammation and allergic reactions. These cells, when triggered by external substances (antigens), can cause inflammation and produce various chemicals that lead to allergic symptoms. Central to this process is a protein called spleen tyrosine kinase (Syk), which, when [...]

by mandy | Mar 15, 2024 | Evidence Based, Inflammation, Nutrients, Nutrition, Science-Based, Wellness
Iron overload is a condition traditionally linked to a host of health issues ranging from liver damage and diabetes to heart problems and distinct changes in skin coloration in severe cases. Symptoms of moderate iron overload include fatigue, joint pain, mood swings, hair loss, and more. The repercussions extend to an elevated risk of neurodegenerative [...]

by mandy | Jan 30, 2024 | Digestive/Urinary, Evidence Based, Inflammation, Nutrients, Nutrition, Science-Based, Supplements, Wellness
https://youtu.be/27PGnCssR8I?si=d40pjClWGwEYe7UG UTI and Chronic UTI- Vitamin D Connection In my recent video, I talk about some of the root causes of IC and chronic UTI, and this includes a protein called cathelicidin. This becomes very important in the context of chronic UTI and biofilm infections associated with IC and Bladder Pain syndrome. Urinary Tract Infections [...]

by mandy | Jan 12, 2024 | Detox, Digestive/Urinary, Evidence Based, Inflammation, Nutrition, Science-Based, Uncategorized, Wellness
In the intricate dance of human biology, trace elements play crucial yet often understated roles. Among them, copper stands out for its significant impact on reproductive health. This essential nutrient, though required in minute quantities, plays a pivotal role in various biological processes critical to reproductive function. In this blog, we will dive into the [...]

by mandy | Dec 14, 2023 | Diet, Evidence Based, Inflammation, Nutrition
https://youtu.be/U9ik9yDwvRk?si=kHknHWW4b95Mhsdg Introduction to AGEs Advanced Glycation End Products, commonly referred to as AGEs, are compounds that naturally form in the body when sugars react with proteins or lipids without the assistance of enzymes. This reaction, known as glycation, occurs slowly over time and is a part of the body's metabolic processes. However, AGEs can also [...]

by mandy | Nov 29, 2023 | Evidence Based, Nutrients, Nutrients, Nutrition, Science-Based
Calcium, a fundamental mineral in the human body, plays a pivotal role in maintaining overall health. Primarily known for its contribution to skeletal structure and bone strength, calcium serves as a cornerstone mineral for an array of bodily functions essential for optimal well-being. Below are just some of the roles Calcium plays in the body. [...]







