

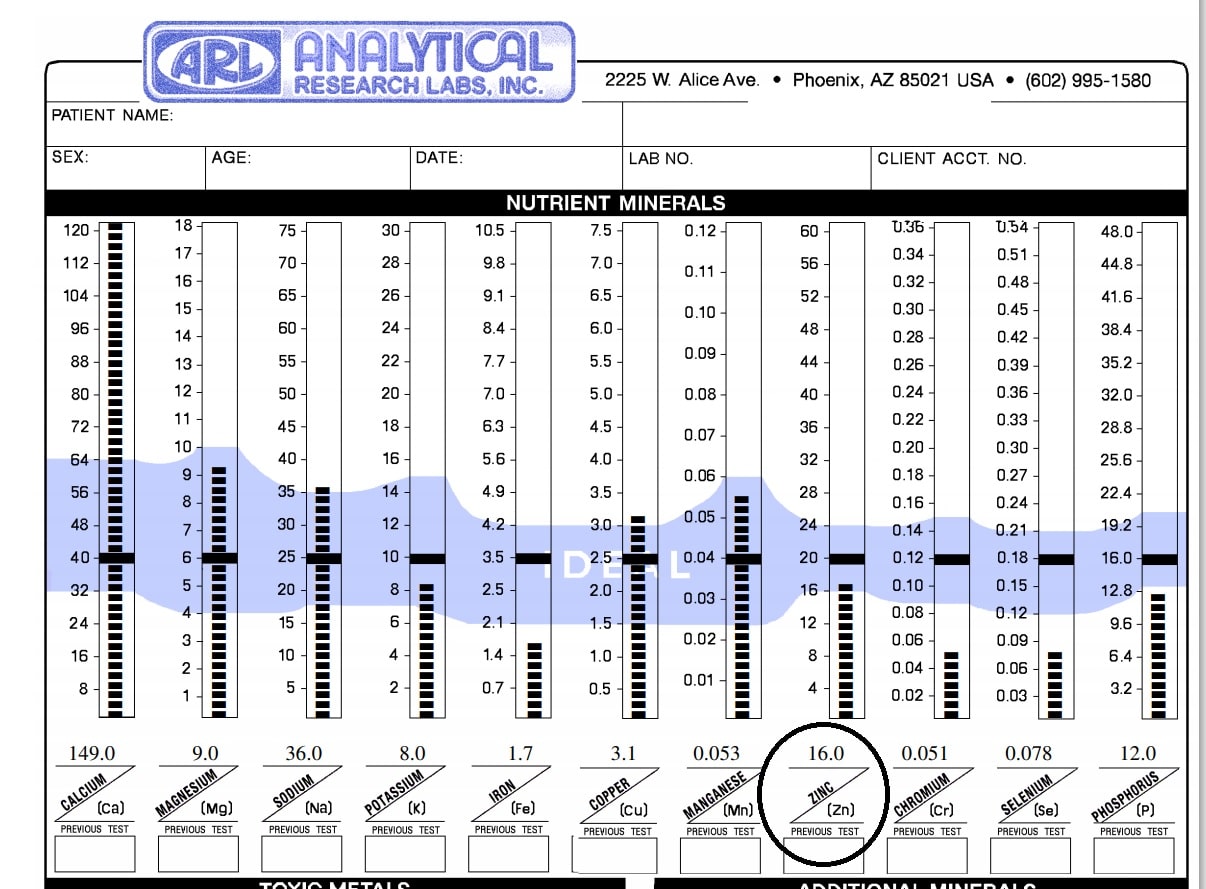
Zinc- a truly important mineral
Zinc is a very interesting mineral. It is also one I commonly see low in hair tissue mineral analysis (HTMA). It plays an important role in facilitating hundreds of biochemical reactions. Due to its role in enzymatic function, it can impact metabolic pathways such as carbohydrate, protein, nucleic acid, and lipid metabolism. It is also [...]
Thiamine Deficiency, Insulin Resistance and Diabetes
The relationship between thiamine and diabetes mellitus (DM) has been reported in the literature (Luong & Nguyen, 2012). Thiamine acts as a coenzyme for transketolase (Tk) and for the pyruvate dehydrogenase (PDH) and α-ketoglutarate dehydrogenase complexes. These enzymes play a fundamental role for intracellular glucose metabolism by increasing Krebs cycle activity (Luong & Nguyen, 2012). [...]

